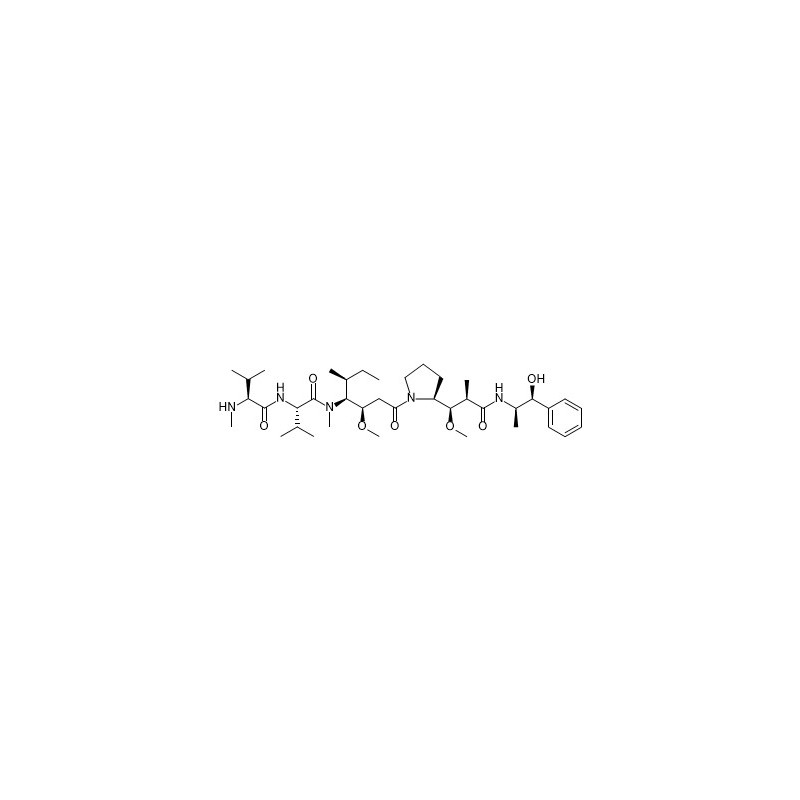

ALB Materials Inc is a supplier of reference standards for over ten years. We keep thousands of inventories and can ship out products within one week after QC test when customer place an order. We supply MMAE (Monomethyl Auristatin E)(CAS#474645-27-7) with high quality for research purpose.
| Item No. | ADCs-T0004 |
| Product Name | MMAE (Monomethyl Auristatin E) |
| CAS Number | 474645-27-7 |
| Molecular Formula | C39H67N5O7 |
| Molecular Weight | 717.98 |
| Purity | 95% |
| Pack Size | 20mg |
| Certification|Grade | NMR|HPLC |
| Form | |
| Type |
For bulk quantity, you can send us your inquiry for a quote.
Data sheet